Medtronic Stealth AXiS Surgical System Gains CE Mark for Spine and Cranial Procedures
Medtronic has received CE Mark approval for its Stealth AXiS surgical system, enabling its use across Europe for spine and cranial procedures. The approval follows earlier U.S. FDA clearances.
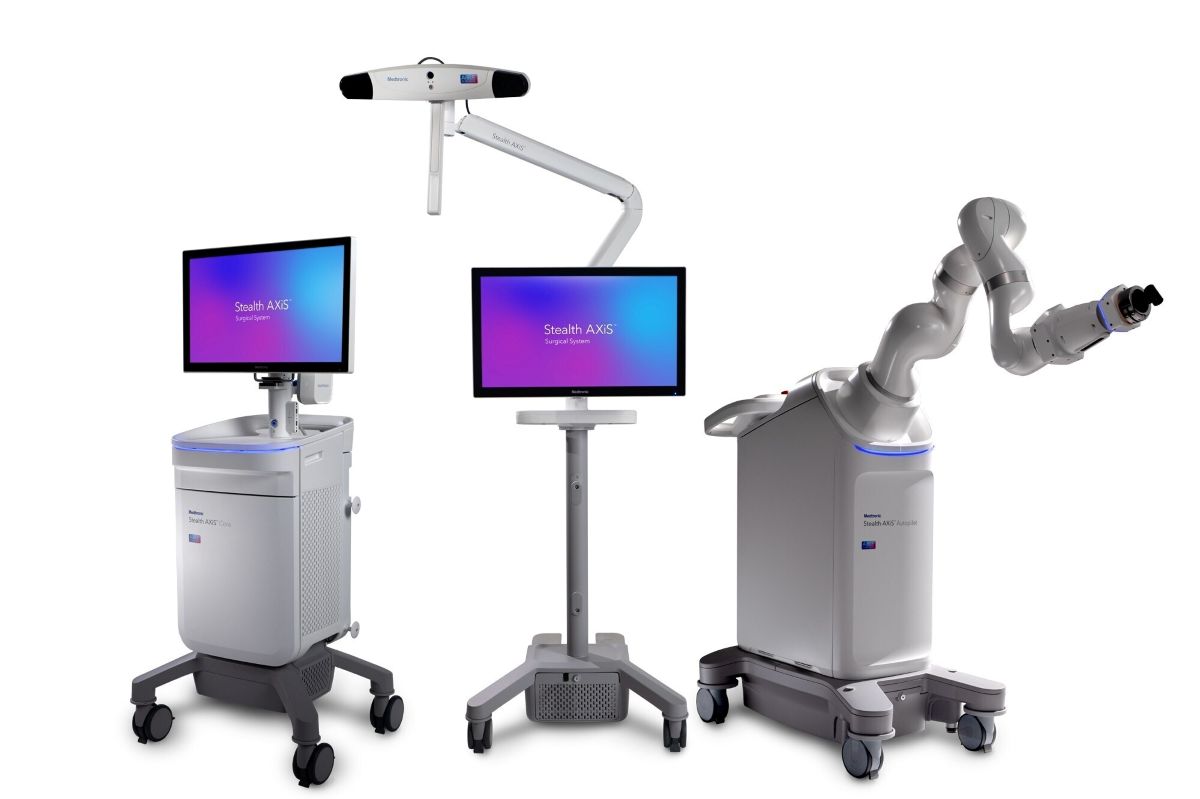
The system integrates surgical planning, navigation and robotics into a single platform designed to support different clinical workflows. It allows hospitals to adopt features based on their needs while maintaining flexibility for future upgrades.
A key feature is LiveAlign segmental tracking, which provides real time visualization of anatomical movement and patient alignment during spine procedures. The platform is part of Medtronic’s AiBLE ecosystem, connecting pre operative, intra operative and post operative processes.
The system also uses AI for advanced planning and visualization. In cranial procedures, it includes automatic tractography to create patient specific brain maps and highlight critical neural pathways.
The CE Mark under the EU Medical Device Regulation supports wider adoption of the system across Europe and reflects Medtronic’s focus on integrated surgical technologies.














Leave a comment